|
For example, calcium combines with carbon to make calcium carbonate, which makes up limestone, marble and seashells. Although they're less reactive than alkali metals, they're not usually found alone in nature. These elements have two valence electrons, which they yield in chemical reactions. Sodium gas fills streetlights, while sodium liquid is used to transfer heat in certain types of nuclear reactors.Īlkaline earth metals (group 2 or IIA) include magnesium, calcium and barium among others. They have one valence electron (or outermost electron that's farthest from the nucleus), which they give up in chemical reactions. 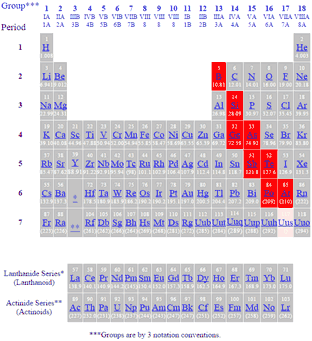
They get their name from their chemical reactions with water in which they produce highly alkaline substances such as sodium hydroxide or lye. For example, hydrogen looks like it should belong to group 1, the alkali metals, but it actually prefers the company of nonmetals.Īlkali metals (group 1 or IA) such as lithium, sodium and potassium, are highly reactive and aren't usually found freely in nature. Let's look at some of these element cliques and remember, sometimes group members are spread around the table, not necessarily in one neat column. 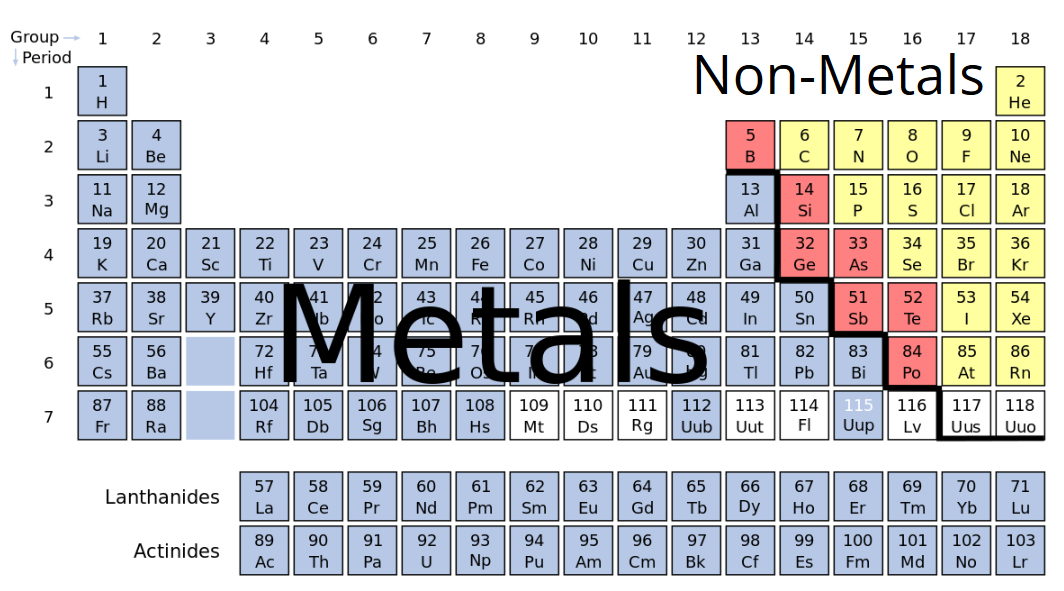
The remaining elements are metalloids, which share properties of both metals and nonmetals. About 80 percent of the elements are metals (shiny elements that conduct heat and electricity well), and 15 percent of the elements are nonmetals (poor conductors of heat and electricity). Groups indicate elements with similar chemical and physical properties. The columns that comprise the periodic table are called groups - 18 in total.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
